Keloid in a Patient with Albinism
Jonel May Breytenbach, FC Plast Surg
Kenneth Segwapa, FC Plast Surg
SUMMARY
Keloids are limited to humans and although the exact prevalence is unknown, they have been reported in 5%-16% of Africans. Individuals with darker pigmentation are more susceptible to keloid formation, especially Africans and Asians. According to the literature, Caucasians are least effected and there has been only a few reports of albino patients with keloids. The author identified a patient with albinism who presented with a keloid to his right ear after trauma at the Dr George Mukhari Academic Hospital in Garankuwa, South Africa. The keloid was resected and sent for pathological evaluation to determine differences in collagen formation. The specimen was compared with tissue from a similar African patient with a keloid due to trauma on the left ear. Both keloids were noticed six months after the sustained trauma.
Keywords: Keloid; Albinism; Histology
INTRODUCTION
Keloids are collagenous lesions that extend beyond the original area of injury and are the result of abnormal wound healing.1 The etiology of keloid formation is unknown,2 but it is believed to be from aberrant wound healing3 caused by excessive fibroblast participation and collagen deposition.3 The highest incidence of keloids has been reported to occur between the first and third decade of life4 and the African population is most affected, with an incidence of 6%-16%.3 Many articles report that patients with albinism are spared from developing keloids3,5-9 and plastic surgery text books generally assert that keloids in patients with albinism have not been reported. Only one epidemiologic publication10 has reported a keloid prevalence rate of 7.8% in people with albinism in Kenya and Tanzania. To my knowledge, there is only one reported case of keloidalis nuchae in a patient with albinism who is from Mali.11
Here we report the first case of an earlobe keloid in a patient with albinism and discuss routine histologic findings compared with an earlobe keloid from an African non-albino patient.
CASE REPORT
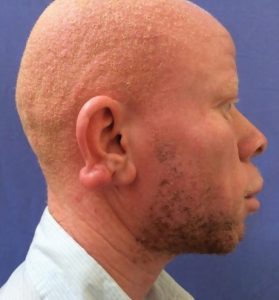
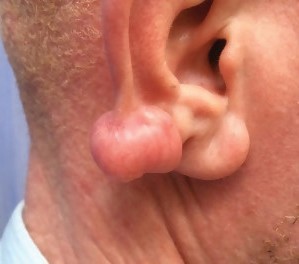
A 26-year-old patient with albinism presented to the plastic surgery clinic at the Dr George Mukhari Academic Hospital in Garankuwa, South Africa, with an earlobe keloid due to a traumatic injury to the ear; the patient had been stabbed with a bottle six months earlier (Fig. 1). He presented with a firm, hard, non-fluctuant, nodular mass on his right ear (1.8 cm x 1.2 cm x 1.2 cm). The lesion, which extended beyond the borders of the original injury, was excised under local anesthetic. The specimen was sent for pathological examination and the patient received radiotherapy. The histological specimen was then compared with an 18-year-old African patient who developed a firm, hard, non-fluctuant, nodular keloid mass on the left ear that stemmed from an ear-piercing six months earlier (2 cm x 2 cm x 0.7 cm). As both histological specimens were diagnosed as keloids, we decided to compare histologic findings to see whether the patient with albinism had a different composition of collagen.
Both parents of the patient with albinism are black of South African descent. He has a pigmented beard and brown eyes and therefore falls under the oculo-cutaneous albinism type 3 with a defect in the TYRP1 gene, usually affecting dark-skinned people and common in black South Africans, who exhibit features of reddish-brown skin, reddish hair, and hazel or brown eyes.
Figure 1: Earlobe keloid on patient with oculo-cutaneous albinism type 3.
RESULTS
The results of the comparison of the histological specimens are shown in Table 1. The albino patient was Fitzpatrick skin type I and the non-albino patient was Fitzpatrick skin type VI.
Table 1. Histological results comparing keloid specimens of the albino and non-albino patients
Fitzpatrick I Albino patient
Non-pigmented, atrophic epidermis
Broad, irregular, thick, homogenous masses of collagen that are acellular, broad coiled bundles of collagen
Focal chronic inflammatory infiltrate
Keloidal features starting mid-dermis
Fitzpatrick VI African patient
Epidermis with adnexal structures (hair follicles and sebaceous glands); epithelium of normal thickness
Proliferation of connective tissue extends in whorls and bundles up to the deepest excision margin
Focal chronic inflammatory infiltrate
Keloidal features starting lower down in the dermis
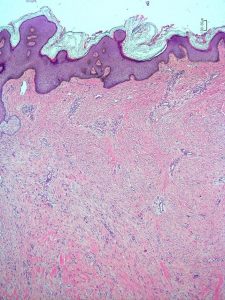
Figure 2: H&E staining of keloid from albino patient (A) and African patient (B). Magnification 4x.

Thin epidermis Keloid starting mid-dermis
Normal epidermis Keloid starting lower in dermis
Examination under microscopy revealed few differences in appearance. The keloid from the albino patient displayed an atrophic epidermis with irregular, thick, homogenous collagen fibres that was acellular and the keloidal features started mid-dermis (Fig. 2A). The non-albino African control patient had normal epidermal thickness and the keloidal features of irregularly oriented collagen whorls started lower in the dermis (Fig. 2B). Due to the observed differences of the dermal structure, the specimens were stained to investigate possible variabilities in collagen types. Masson’s trichrome staining selectively stains for muscle, collagen fibers, fibrin, and erythrocytes and was used here to identify collagen fibers, which stain blue (Figs. 3A and B).
Picrosirius stain (Merck, Garankuwa, SA) is a connective tissue stain that is used to visualize type I and type III collagen in skin. Collagen can be visualized under normal microscopy light or under polarized light, which results in birefringence of the collagen fibers that helps to distinguish type I and type III collagen. Type I collagen shows thicker bundles with yellow-orange birefringence and type III shows green birefringence (Figs. 6 and 7).
Using H&E, Masson’s trichrome (Merck, Garankuwa, SA), and picrosirius red staining techniques, no significant distinguishing features between the specimen from the patient with albinism and from the control patient were found.
DISCUSSION
This paper reports on the occurrence of an earlobe keloid in a patent with albinism and may be the first such reported case from South Africa. In a research letter, Kiprono et al. (2015)10 presented epidemiologic data of keloid patients with albinism from Kenya and Tanzania, with a keloid prevalence of 7.8% compared with 8.3% in non-albinism patients with normal skin pigment. Unfortunately, that report did not further elaborate on keloids in patients with albinism and went largely unnoticed in the current keloid literature, as did Frikh and Boui’s case report.11 Both descriptions failed to classify the albino type of their patients.
In this study, we had the opportunity to compare the histology of two earlobe keloids from two patients of similar age, one of whom had oculo-cutaneous albinism. There were no significant differences between the two, although the patient with albinism showed a more advanced and mature lesion: The collagen fibers were thicker and the epidermis atrophic, suggesting a more aggressive keloid growth. However, this difference could be attributed to the severity of the trauma, which could have evoked a more pronounced inflammatory response.
CONCLUSION
This report confirms that keloids do exist in patients with albinism. However, the histological appearance was not different from a similar lesion in a patient of comparable age with normal skin pigmentation. As more patients with albinism and keloids are identified, histological, genetic, and molecular analyses should be performed to study whether there are differences in keloid formation in this population.
Disclosure
The author has no financial interests to declare.
Grant Information
The author declares that no grants were involved in supporting this work.
REFERENCES
1. Andrews JP, Marttala J, Macarak E, Rosenbloom J, Uitto J. Keloids: the paradigm of skin fibrosis – pathomechanisms and treatment. Matrix Biol. 2016; 51:37-46.
2. Gauglitz GG, Korting HC, Pavicic T, Ruzicka T, Jeschke M. Hypertrophic scarring and keloids: pathomechanisms and current and emerging treatment strategies. Mol Med. 2011; 17:113-25.
3. Huang C, Murphy GF, Akaishi S, Ogawa R. Keloids and hypertrophic scars: update and future directions. Plast Reconstr Surg Glob Open. 2013;1:e25.
4. Bran GM, Goessler UR, Hoffman K, Riedel F, Sadick H. Keloids: current concepts of pathogenesis (review). Int J Mol Med. 2009; 24:283-93.
5. Edriss AS, Mesták J. Management of keloid and hypertrophic scars. Ann Burns Fire Disasters. 2005; 18:202-10.
6. Chike-Obi CJ, Cole PD, Brissett AE. Keloids: pathogenesis, clinical features, and management. Semin Plast Surg. 2009; 23:178-84.
7. Kelly PA. Update on the management of keloids. Seminars in Cutaneous Medicine and Surgery. 2009; 28:71-6.
8. Viera MH, Vivas AC, Berman B. Update on keloid management: clinical and basic science advances. Adv Wound Care (New Rochelle). 2012; 1:200-6.
9. Song C. Hypertrophic scars and keloids in surgery: current concepts. Ann Plast Surg. 2014;73 Suppl 1:S108-18.
10. Kiprono SK, Chaula BM, Masenga JE, Muchunu JW, Mavura DR, Moehrle M. Epidemiology of keloids in normally pigmented Africans and African people with albinism: population-based cross-sectional survey. Br J Dermatol. 2015; 173:852-4.
11. Frikh R, Boui M. Albinisme et folliculite fibrosante de la nuque chez un homme d’ascendance africaine. Annals de dermatologie et de venereologie. 2015; 142:690-691.
LEGENDS: FIGURESLEGENDS: FIGURES
Fig. 1 Keloid on the right ear of a patient with albinism
Fig 2A Histology specimen of patient with albinism under 4 x magnification
Fig 2B Histology specimen of African patient under 4 x magnification
Fig. 3A Masson’s trichrome stain for the patient with albinism, staining the collagen blue 4 x magnification
Fig. 3B Masson’s trichrome stain for the African patient 4 x magnification
Fig. 4A Picrosirius stain for the patient with albinism. Yellow and red birefringence is seen indicating collagen type 1 as well as a lesser amount of collagen type III (green birefringence)
Fig. 4B Picrosirius stain for the patient without albinism. Similar patterns of staining are seen in Fig. 4A
LEGENDS: TABLE
Table 1. Histological results from the keloid specimens from albino and non-albino patients
